Peer Reviewed
Cutaneous Acanthamoeba Infection in a Patient With Multiple Sclerosis on Fingolimod
Authors:
Eric C. Schmidgal, MD; John Jackson, MD; and Doyle Hansen, MD
San Diego, California
Citation:
Schmidgal EC, Jackson J, Hansen D. Cutaneous acanthamoeba infection in a patient with multiple sclerosis on fingolimod [published online November 27, 2018]. Neurology Consultant.
A 66-year-old woman with multiple medical problems, including relapsing-remitting multiple sclerosis (MS) treated with fingolimod, originally presented to her primary care provider (PCP) for a suspected skin infection of her right upper extremity.
The patient had a firm 1.5-cm nodule with a boggy center on her right forearm, and she had been concerned that a potential insect bite or thorn injury while gardening had become infected. The PCP had biopsied the lesion, the results of which had shown a granulomatous and suppurative dermatitis that was negative for any specific organisms. She had been treated with doxycycline without resolution or changes to the nodule and then had been referred to our dermatology practice for further workup.
Physical examination. Upon presentation, the nodule remained as described above, without growth or ulceration. The patient was afebrile, with no adenopathy, and her physical examination findings and vital signs were otherwise normal.
Diagnostic tests. A second biopsy was performed; at follow-up approximately 4 months after her initial presentation to primary care, the site of the original nodule had evolved into a 3.5-cm ulceration with surrounding erythema. A wick was placed at the second biopsy site to promote drainage (Figure 1).
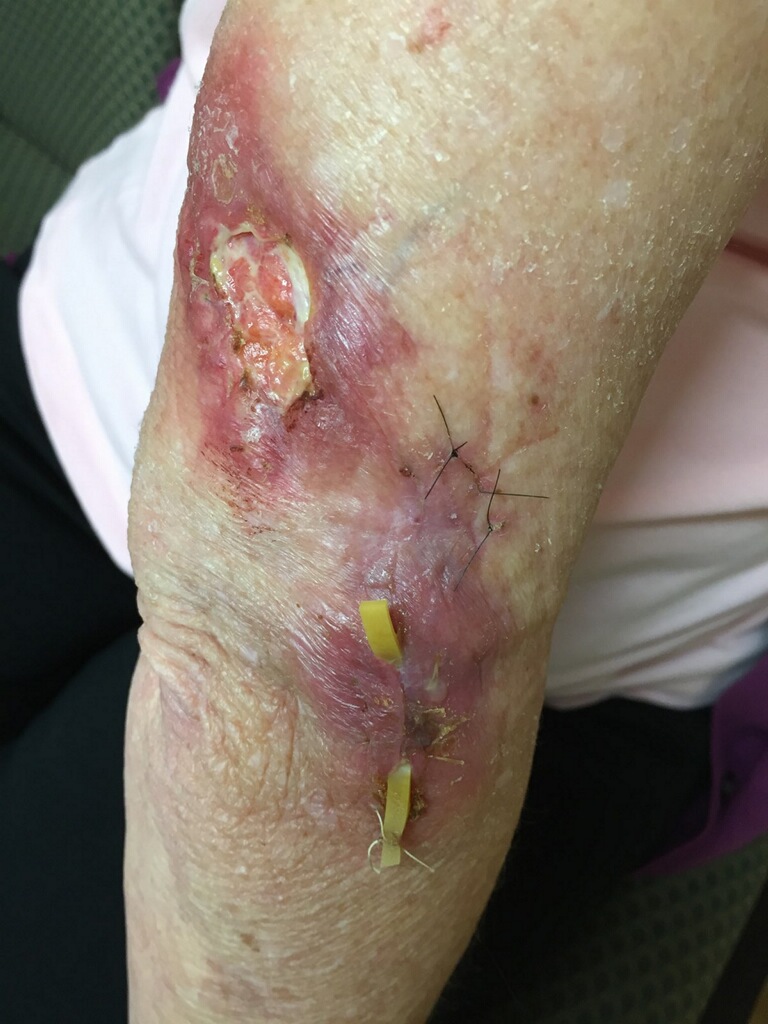
Figure 1. Approximately 4 months after initial presentation, the nodule had become a 3.5-cm ulceration with surrounding erythema; 2 sutures from the original biopsy are visible, as well as a wick placed at the subsequent biopsy site to promote drainage.
The second biopsy of the lesion was sent for hematoxylin and eosin (H&E) staining and tissue cultures, including atypical mycobacteria and opportunistic fungi (Figures 2 and 3).
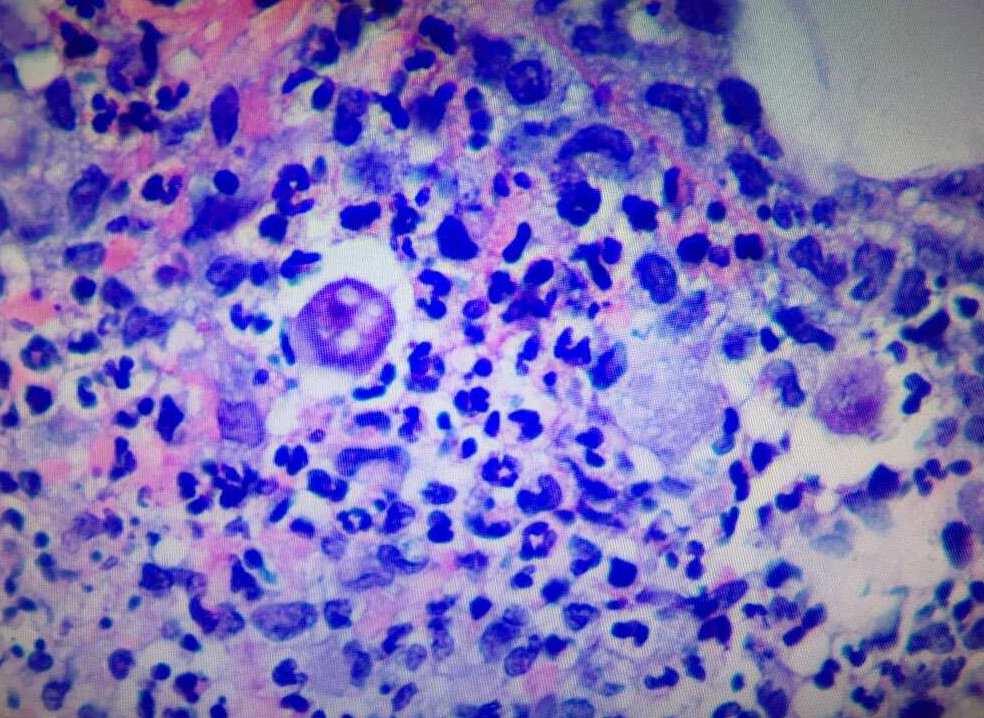

Figure 2. H&E staining of the biopsy specimen at ×20 magnification showed mixed inflammatory infiltrate with lymphocytes, histiocytes, and polymorphic leukocytes.
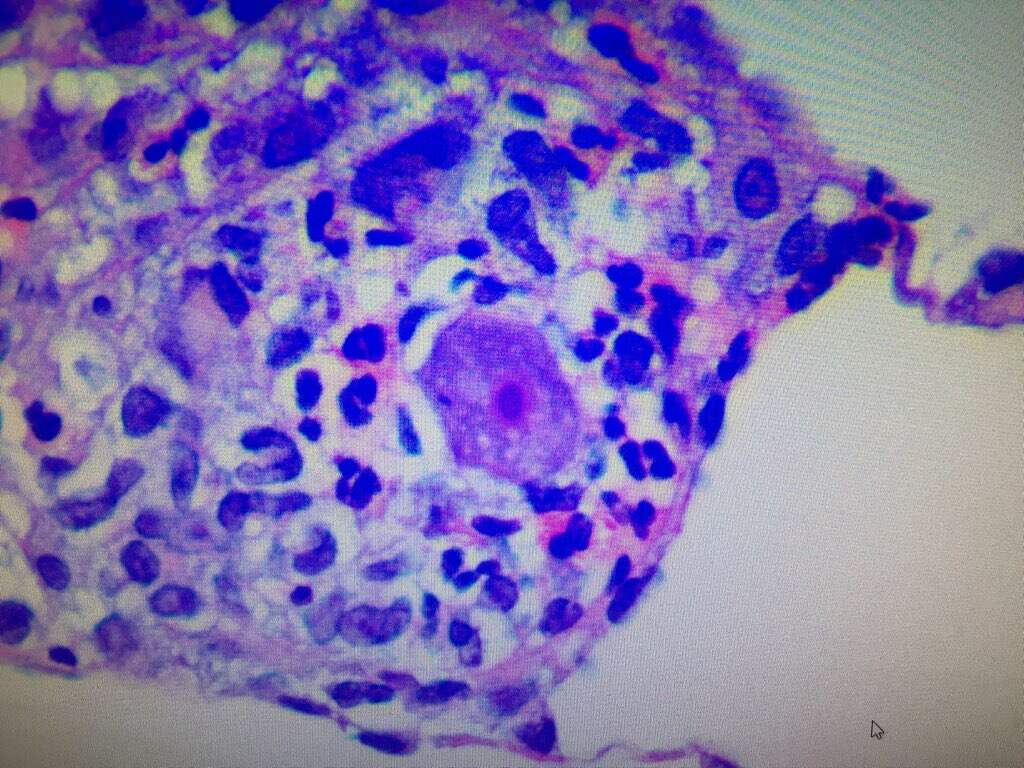
Figure 3. H&E staining of same biopsy at ×20 magnification. Note the trophozoite with a agenta-colored nucleus and prominent nucleoli at center of image.
Special staining was negative for acid-fast bacilli and fungal organisms; however, scattered organisms present on the tissue were concerning for ameba. A tentative diagnosis of cutaneous amebiasis was made, and the patient was started on oral metronidazole, 500 mg 3 times a day, and was referred to an infectious disease specialist for additional recommendations.
For supplementary testing, microscopic photographs and a formalin-fixed tissue slide were sent to the Centers for Disease Control and Prevention (CDC) in Atlanta, Georgia. The CDC was able to confirm the diagnosis of cutaneous acanthamebiasis and isolated Acanthamoeba healyi using indirect immunofluorescence assay and polymerase chain reaction. The CDC recommended intravenous treatment if there concern existed for disseminated granulomatous amebic encephalitis.
Based on confirmatory testing by the CDC and the recommendations from the infectious disease specialist, the patient’s metronidazole was switched to oral fluconazole, and she continued to receive routine wound care with debridement and dressing changes. Magnetic resonance imaging of the brain and cervical spine revealed stable MS plaques without evidence of disseminated acanthamebiasis. Because the patient remained asymptomatic and without evidence for disseminated disease, intravenous medications were not initiated.
Outcome of the case. After discussion with the patient and her neurologist, it was decided to discontinue her fingolimod. Her cutaneous infection continued to heal well with routine wound care, fluconazole, and discontinuation of fingolimod. After complete resolution of the acanthamebiasis, she eventually resumed fingolimod therapy with close follow-up from her neurologist and dermatologist. She has since remained free of cutaneous findings.
Discussion. Fingolimod is a disease-modifying drug used to treat relapsing/remitting-type MS. It is the first oral medication to receive approval from the Food and Drug Administration to treat this type of MS. The drug interacts with sphingosine phosphate-1 receptors (S1P1, S1P3, S1P4, and S1P5), preventing lymphocytes from passing out of lymphoid tissue. Fingolimod initially activates lymphocyte S1P1 via receptor binding and eventually induces S1P1 down-regulation. Inhibiting lymphocyte (especially the subsets responsible in MS) exiting from lymphoid tissues reduces autoreactive lymphocyte infiltration into the central nervous system (CNS).1
Peripheral lymphopenia occurring in patients on fingolimod initially raised concern about the potential for opportunistic infection; however, this has not been borne out in clinical trials of the drug. Fingolimod-treated groups had the same number of infections as control groups.2,3 More recent data have shown, however, a slight increase in varicella-zoster virus infections in patients on fingolimod vs placebo.4
Acanthamoeba is a recognized opportunistic infectious agent in the immunocompromised host. Ameba belonging to the genus Acanthamoeba have been isolated from soil, water, vegetables, fish, and birds.5 They have been cultured from multiple body sites on humans, including skin, eyes, ears, throat, nasal and sinus cavities, and lungs.6 Acanthamoeba exist in both dormant cyst and metabolically active trophozoite forms. During the trophozoite stage, which is the infectious form, acanthamoeba feed on organic particles as well as other microbes and reproduce if conditions (eg, pH, temperature, food) are favorable. Exposure to harsher environments causes transformation into a double-walled cyst form.5
Cutaneous acanthamoeba infection without CNS involvement have been treated successfully with numerous medications including pentamidine isethionate, sulfadiazine, flucytosine, and fluconazole or itraconazole.7 Topical treatments can include chlorhexidine and ketoconazole cream in appropriate nonimmunocompromised patients. Fluconazole, as used in our patient’s case, works by inhibiting the fungal cytochrome P450 enzyme 14α-demethylase. Mammalian demethylase is much less sensitive to fluconazole, making it an effective treatment. Good wound care and debridement are important, as well, for preventing the spread of infection into healthy surrounding tissues.
We propose that although during clinical trials fingolimod had not been shown to predispose patients to opportunistic infection, its use still may be a risk factor for certain opportunistic infections such as acanthamoeba. Patients on fingolimod should be monitored closely for signs of infection, and cutaneous or other infections that do not respond to traditional therapies should prompt further workup, specialist consultation, and consideration of the presence of an opportunistic infection.
- Chun J, Hartung H-P. Mechanism of action of oral fingolimod (FTY720) in multiple sclerosis. Clin Neuropharmacol. 2010;33(2):91-10
- Cohen JA, Barkhof F, Comi G, et al; TRANSFORMS Study Group. Oral fingolimod or intramuscular interferon for relapsing multiple sclerosis. N Engl J Med. 2010;362(5):402-415.
- Kappos L, Radue E-W, O’Connor P, et al; FREEDOMS Study Group. A placebo-controlled trial of oral fingolimod in relapsing multiple sclerosis. N Engl J Med. 2010;362(5):387-401.
- Arvin AM, Wolinsky JS, Kappos L, et al. Varicella-zoster virus infections in patients treated with fingolimod: risk assessment and consensus recommendations for management. JAMA Neurol. 2015;72(1):31-39.
- Bottone EJ. Free-living amebas of the genera Acanthamoeba and Naegleria: an overview and basic microbiologic correlates. Mt Sinai J Med. 1993;60(4):260-270.
- Tan B, Weldon-Linne CM, Rhone DP, Penning CL, Visvesvara GS. Acanthamoeba infection presenting as skin lesions in a patient with the acquired immunodeficiency syndrome. Arch Pathol Lab Med. 1993;117(10):1043-104
- Siddiqui, R, Aqeel Y, Khan NA. The development of drugs against Acanthamoeba infections. Antimicrob Agents Chemother. 2016;60(11):6441-6450.


